 These chemical reactions involve the movement of electrons from one species to another. You'll then identify unknown metal electrodes using known standard potentials and determine the magnitude of the voltage produced.Įlectrochemistry is a branch of chemistry that studies the relationship between electrical energy and a chemical change. In this lab, you will construct a galvanic cell using two metal electrodes and measure the potential produced as the oxidation and reduction reactions occur. If the metals have different standard potentials, like silver and magnesium, the voltage produced will be high. So, if two metals with similar standard electrode potentials are used in the galvanic cell, like lead and tin, for example, the magnitude of voltage produced will be low. The magnitude of the voltage produced is equal to the standard potential of the cathode minus the standard potential of the anode. As the electrons travel between the two electrodes, an electric potential is measured. In a galvanic cell, electrons flow from the anode to the cathode, thus the anode is the metal with the lower standard potential, and the cathode is the metal with the higher standard potential. Electrons always flow from the metal with the lower standard potential to the metal with the greater standard potential. A more negative electrode potential means that the metal has a higher tendency to lose electrons. 
So how do we know which metal acts as the anode and which acts as the cathode? We can use the standard electrode potential of the metal, which is a measure of the metal’s tendency to lose electrons. To help you distinguish between these two labels, remember the phrase 'red cat,' since reduction occurs at the cathode. The cathode is the electrode that gains electrons and is reduced. The anode is the electrode that gives up electrons in the reaction or is oxidized. The electrodes are connected by an external circuit, which the electrons travel through during the reaction. The solutions are connected by a salt bridge, which enables the flow of ions and maintains the charge neutrality of the solutions. The solutions consist of the ions needed for the half-reactions, either the oxidation or reduction reaction, that occur at each electrode. 
First, there are two metal electrodes, each of which is immersed into a different electrolyte solution. Some electrochemical reactions occur spontaneously, which we can study in a galvanic cell. To help you distinguish between the two, remember the phrase 'oil rig,' which stands for oxidation is losing, reduction is gaining. When it gains electrons, we say that it has undergone reduction or has been reduced. When a substance loses electrons, we say that it has undergone oxidation or that it has been oxidized. 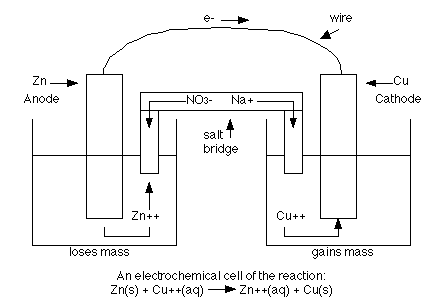
If you measure the voltage under load, it will also be affect by the nature of the salt bridge.An electrochemical reaction is a process where electrons are transferred between two substances, with one losing electrons and the other gaining them. If ion transport along the salt bridge is the rate-determining step, it will affect the current (current is movement of charge per time). The voltage and the current depend on what load you attach to the voltaic cell, and properties of the cell itself. A similar (but reversed) situation is found in the cathodic cell, where $\ce$ in making the salt bridge have any effects on voltage/current output of the cell? why? 
In order to maintain neutrality, the negatively charged ions in the salt bridge will migrate into the anodic half cell. The electrons move through the wire (and your device, which I haven't included in the diagram), leaving the unbalanced positive charge in this vessel. The oxidation reaction that occurs at the anode generates electrons and positively charged ions. The electrons flow from the anode to the cathode. The purpose of a salt bridge is not to move electrons from the electrolyte, rather it's to maintain charge balance because the electrons are moving from one-half cell to the other. There's another question related to salt bridges on this site. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
